Severe thalassemia is a genetic blood disorder. Understanding the risk factors of severe thalassemia, early diagnosis through hemoglobin panel testing, and proper thalassemia treatment helps manage complications, reduce iron overload, and improve long-term health outcomes and quality of life.
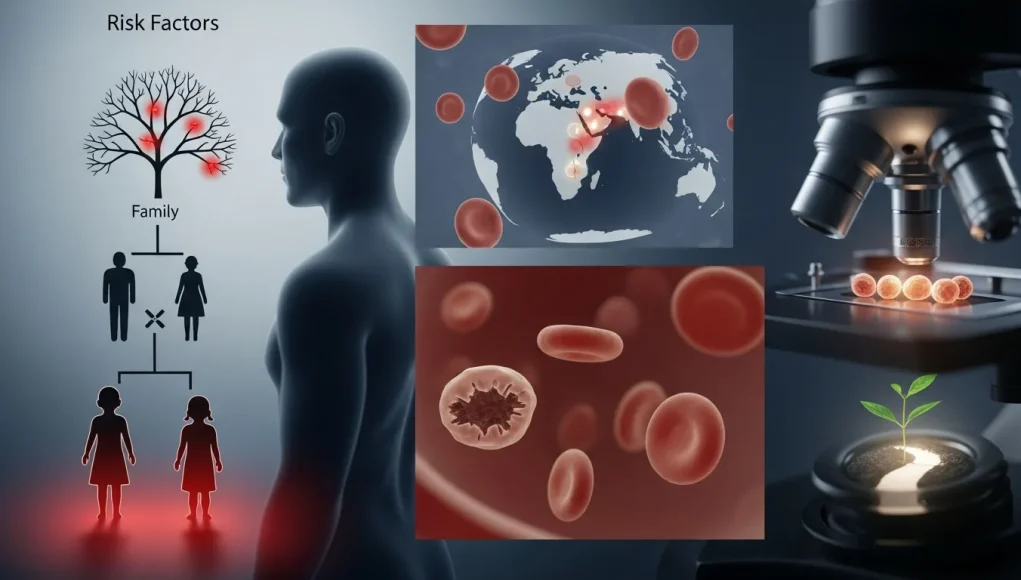
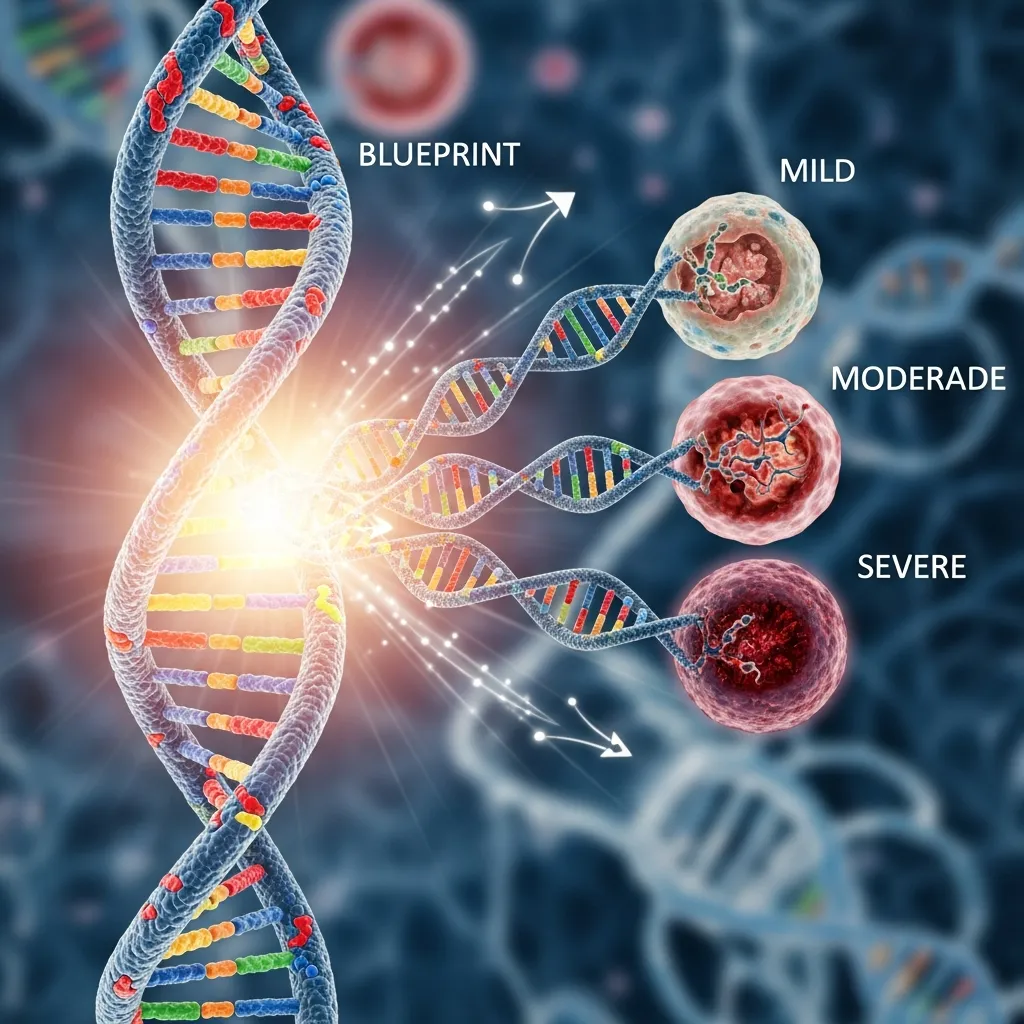 The primary risk factor for any form of thalassemia is the genetic mutations inherited from parents. The severity of the disease is almost entirely determined by which genes are affected, how many are missing, and whether the mutation is present in one or both parents. This is why understanding the risk factors of severe thalassemia is essential for early detection, genetic counseling, and prevention planning.
The primary risk factor for any form of thalassemia is the genetic mutations inherited from parents. The severity of the disease is almost entirely determined by which genes are affected, how many are missing, and whether the mutation is present in one or both parents. This is why understanding the risk factors of severe thalassemia is essential for early detection, genetic counseling, and prevention planning.
In many cases, individuals may be carriers of the gene without showing any symptoms. However, when two carriers have a child, the risk of severe disease increases significantly. This makes family history and genetic screening a key part of thalassemia diagnosis and prevention strategies.
Beta Thalassemia Mutations
In beta thalassemia, the body struggles to produce beta-globin chains, which are essential components of hemoglobin. When one gene is affected, symptoms may be mild or absent. However, if both beta-globin genes are mutated, the condition becomes beta thalassemia major, the most severe form of the disease.
Patients with beta thalassemia major often require lifelong blood transfusion therapy to survive. Without regular treatment, severe anemia can lead to growth delays, heart complications, and a significantly increased risk of early mortality. Long-term management also includes monitoring through regular hemoglobin panel testing and iron level evaluation to prevent complications such as iron overload.
Alpha Thalassemia and Hydrops Fetalis
Alpha thalassemia involves four genes responsible for alpha-globin production. The severity depends on how many of these genes are missing or defective. If all four genes are absent, it leads to a condition called hydrops fetalis, which is usually fatal before or shortly after birth.
In such high-risk cases, early detection during pregnancy is extremely important. Recognizing alpha thalassemia symptoms during pregnancy allows doctors to provide genetic counseling, prenatal monitoring, and family planning guidance. Early screening helps reduce risks and prepares parents for possible medical interventions.
The Role of Ethnicity
Genetics and ancestry play a significant role in the prevalence of thalassemia. People of Mediterranean, South Asian, Southeast Asian, and Middle Eastern descent are statistically at a higher risk of carrying thalassemia gene mutations. This makes population-based screening programs highly important in these regions.
Because of this geographic distribution, pre-marital and pre-conception screening has become an essential preventive measure in many countries. Identifying carriers early helps reduce the chance of passing severe forms of the disease to children and supports better long-term public health outcomes.
The Iron Overload Dilemma
For individuals with severe thalassemia, regular blood transfusions are a life-saving treatment that helps maintain adequate hemoglobin levels and ensures proper oxygen delivery to the body. However, while essential for survival, these transfusions introduce one of the most serious secondary complications: iron overload. This condition is a major concern when discussing the risk factors of severe thalassemia, as it significantly affects long-term organ health.
Why Iron Overload Happens
The human body does not have a natural mechanism to eliminate excess iron. Under normal conditions, iron levels are carefully balanced through diet and absorption. However, each unit of transfused blood contains approximately 200–250 mg of iron.
In patients with severe thalassemia who require frequent transfusions—often 20 or more per year—this iron gradually accumulates in the body. Over time, this buildup becomes toxic and leads to a condition known as transfusion-related iron overload, which can severely damage multiple organs if not properly managed.
Regular monitoring through a hemoglobin panel and iron studies is essential to detect early signs of overload and adjust treatment accordingly.
Impact on the Liver
The liver is the body’s primary storage organ for excess iron. When iron levels exceed its storage capacity, it triggers oxidative stress, inflammation, and gradual tissue damage. This process leads to scarring of the liver tissue, known as fibrosis.
If left untreated, fibrosis can progress to cirrhosis, a severe and irreversible condition that significantly reduces liver function. Early recognition of liver iron overload symptoms, such as abdominal discomfort, fatigue, dark urine, or jaundice, is critical in preventing long-term complications.
Effective management often includes iron chelation therapy, dietary control, and consistent monitoring as part of a comprehensive thalassemia treatment plan.
Endocrine System Risks
Excess iron does not only affect the liver—it also targets the endocrine system, which regulates hormones throughout the body. This makes iron overload one of the most dangerous complications associated with the risk factors of severe thalassemia, especially in long-term patients.
Diabetes Mellitus
Iron accumulation in the pancreas can damage insulin-producing cells, increasing the risk of diabetes mellitus. This condition can make blood sugar control more difficult and require lifelong management.
Growth Retardation
In children and adolescents, iron toxicity may affect the pituitary gland, which plays a key role in growth and development. This can result in delayed growth and puberty, requiring hormonal evaluation and medical support.
Hypothyroidism
Iron buildup in the thyroid gland can reduce hormone production, leading to hypothyroidism. This condition slows metabolism and may cause fatigue, weight gain, and sensitivity to cold.
Cardiovascular Risk Factors
 Cardiovascular complications are among the most serious risk factors of severe thalassemia, and heart failure has historically been the leading cause of death in patients with this condition. What makes this especially dangerous is that cardiac iron overload often develops silently. Many patients do not experience noticeable symptoms until the condition has already progressed to a critical and potentially life-threatening stage.
Cardiovascular complications are among the most serious risk factors of severe thalassemia, and heart failure has historically been the leading cause of death in patients with this condition. What makes this especially dangerous is that cardiac iron overload often develops silently. Many patients do not experience noticeable symptoms until the condition has already progressed to a critical and potentially life-threatening stage.
Regular monitoring and early detection are therefore essential parts of modern thalassemia management, helping to reduce long-term cardiac complications and improve survival rates.
Arrhythmias and Heart Failure
One of the earliest effects of iron accumulation in the heart is disruption of the heart’s electrical system. Iron deposits interfere with normal electrical signaling in the cardiac muscles, which can lead to arrhythmias (irregular heartbeats). These may present as palpitations, dizziness, or short episodes of breathlessness.
As iron continues to build up, the heart muscle becomes progressively weaker and less efficient at pumping blood. This can eventually develop into congestive heart failure, where the heart is unable to supply enough oxygen-rich blood to the body’s organs.
This stage is one of the most severe complications associated with the risk factors of severe thalassemia, and it often requires intensive medical intervention, including iron chelation therapy, medication, and continuous cardiac monitoring.
Monitoring Cardiac Health
Advancements in medical imaging have significantly improved the ability to detect early signs of heart complications in thalassemia patients. One of the most important tools is the T2* MRI scan, which allows doctors to measure iron levels in the heart tissue with high accuracy.
Unlike traditional tests, T2* MRI can detect cardiac iron overload before symptoms become visible. This early detection plays a crucial role in preventing irreversible heart damage and is considered a cornerstone of modern heart care in thalassemia patients.
In addition to imaging, regular hemoglobin panel testing and iron level monitoring are also important for evaluating overall treatment effectiveness and adjusting therapy when needed.
Organ Damage and Skeletal Changes
Severe thalassemia does not only affect the blood—it gradually impacts multiple organs and reshapes the body over time. These complications are considered important risk factors of severe thalassemia, especially in patients who do not receive timely or adequate treatment. Chronic anemia, continuous bone marrow stress, and ongoing red blood cell destruction all contribute to long-term structural and functional damage in the body.
Bone Marrow Expansion
In severe thalassemia, the body continuously tries to compensate for low red blood cell production. As a result, the bone marrow becomes overactive and expands in an attempt to produce more blood cells. This excessive activity places abnormal pressure on the bones.
Over time, this leads to thinning of the bone structure, increased brittleness, and visible deformities. One of the most recognizable effects is facial bone changes, including frontal bossing and malar prominence, which occur due to bone expansion in the skull and cheek area.
These skeletal changes are a direct consequence of chronic anemia and represent one of the long-term structural risk factors of severe thalassemia, particularly when treatment is delayed or inconsistent.
Splenomegaly (Enlarged Spleen)
The spleen plays an important role in filtering damaged or abnormal red blood cells. In severe thalassemia, the constant presence of abnormal cells forces the spleen to work harder than normal. Over time, this leads to splenomegaly, or abnormal enlargement of the spleen.
As the spleen enlarges, it becomes more aggressive in destroying both defective and transfused red blood cells. This creates a harmful cycle where newly transfused blood is rapidly broken down, reducing the effectiveness of treatment and worsening anemia.
This condition is a major form of organ damage in thalassemia and is considered a serious complication. In advanced cases, doctors may recommend a splenectomy (surgical removal of the spleen) to prevent further destruction of red blood cells and improve overall blood stability.
Regular monitoring through a hemoglobin panel and physical examination is essential to detect early signs of spleen enlargement and prevent complications.
Risks in Adulthood and Aging
As medical science advances, more individuals with severe thalassemia are surviving into adulthood, which was once uncommon. While this is a major success in modern healthcare, it also introduces a new set of long-term health challenges known as thalassemia complications in adults. These complications often develop gradually and require lifelong monitoring and management.
One of the most common issues is osteoporosis, which occurs due to continuous bone marrow expansion, hormonal imbalances, and long-term iron overload. Weakened bones increase the risk of fractures, chronic pain, and reduced mobility, significantly affecting quality of life.
Another serious complication is pulmonary hypertension, a condition where blood pressure in the lungs becomes abnormally high. This puts extra strain on the heart and can worsen existing cardiovascular problems, making regular cardiac evaluation essential in adult thalassemia care.
Chronic infections are also more common, especially in patients who have undergone splenectomy (removal of the spleen). Since the spleen plays a key role in immune defense, its absence increases vulnerability to bacterial infections, requiring preventive care and vaccinations.
These adult complications highlight why ongoing monitoring and structured care are essential throughout life in patients with severe thalassemia.
Managing the Risk Factors
 The primary goal of modern medicine is to transform severe thalassemia from a life-threatening childhood disorder into a manageable chronic condition. Effective management focuses on reducing complications, improving quality of life, and extending life expectancy through continuous care.
The primary goal of modern medicine is to transform severe thalassemia from a life-threatening childhood disorder into a manageable chronic condition. Effective management focuses on reducing complications, improving quality of life, and extending life expectancy through continuous care.
Chelation Therapy
Chelation therapy is one of the most critical components in managing the risk factors of severe thalassemia. Patients who receive regular blood transfusions must also undergo iron chelation therapy to remove excess iron from the body.
Medications such as Deferasirox and Desferrioxamine help bind and eliminate iron before it can accumulate in vital organs like the heart, liver, and endocrine glands. Poor adherence to chelation therapy is one of the biggest risk factors for organ failure and reduced survival in thalassemia patients.
Therefore, consistent compliance with chelation treatment is considered one of the most important determinants of long-term health outcomes.
Diagnostic Precision
Accurate and regular monitoring is essential in modern thalassemia care. Following a structured thalassemia test complete guide ensures that patients undergo timely evaluations of hemoglobin levels, serum ferritin, liver function, and heart health.
Routine hemoglobin panel testing also plays a key role in tracking disease progression and treatment effectiveness. Early detection of abnormalities allows doctors to adjust treatment plans before complications become severe.
Consistent diagnostic monitoring significantly reduces the risk factors of severe thalassemia by enabling proactive rather than reactive care.
Emerging Therapies
Recent advancements in medical science are offering new hope for patients with severe thalassemia. Gene therapy and CRISPR-based technologies are at the forefront of these innovations, aiming to correct the underlying genetic defects responsible for the disease.
These advanced approaches represent the future of Advanced Thalassemia Care, with the potential to provide long-term or even permanent solutions. Although still under development and clinical evaluation, they mark a major shift in hematology and personalized medicine.