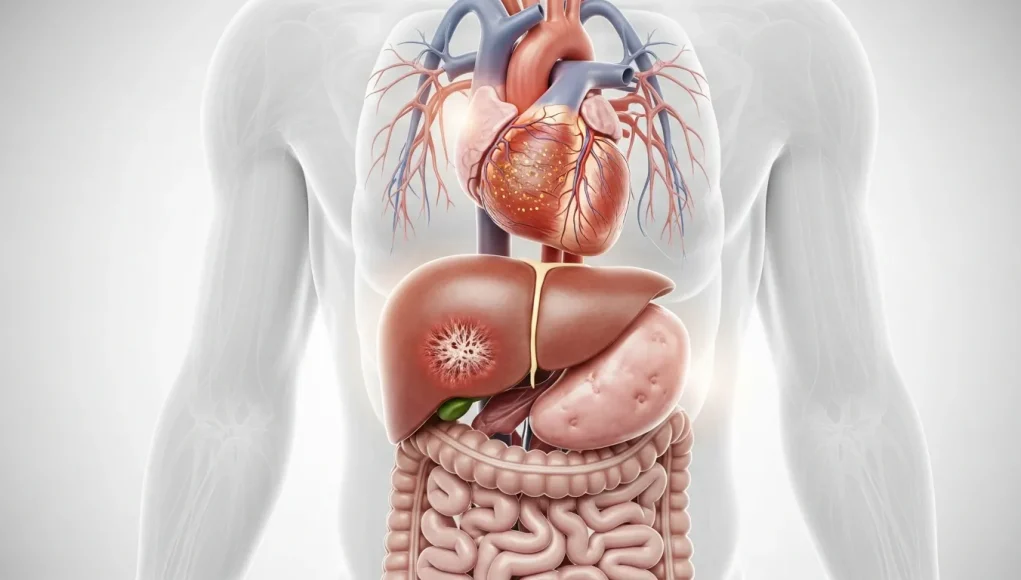
Organ damage in thalassemia occurs due to chronic anemia and iron overload from frequent blood transfusions. It mainly affects vital organs like the heart, liver, and spleen, leading to serious complications if not properly managed and monitored.
The Root Cause: Iron Overload
The most significant driver of tissue damage in patients is a condition known as iron overload. This occurs due to two primary factors: ineffective erythropoiesis and frequent blood transfusions. Ineffective erythropoiesis means the bone marrow struggles to make healthy red blood cells. The body senses this shortage and tries to compensate by absorbing abnormally high amounts of iron from the diet.
Simultaneously, patients with severe forms of the disorder often require regular blood transfusions to survive. Every unit of transfused blood contains a large amount of iron. The human body lacks a natural mechanism to excrete excess iron efficiently. As a result, the mineral builds up rapidly in the bloodstream. When the blood can no longer hold the excess, the free iron begins to deposit directly into tissues and organs.
This toxic accumulation causes severe oxidative stress, which damages cell structures and impairs normal function. To combat this, doctors prescribe chelation therapy. Chelating agents are medications that bind to the excess iron, allowing the body to excrete it safely through urine or stool. Strict adherence to this therapy is the most effective way to prevent long-term tissue toxicity.
Impact on the Heart (Cardiac Complications)
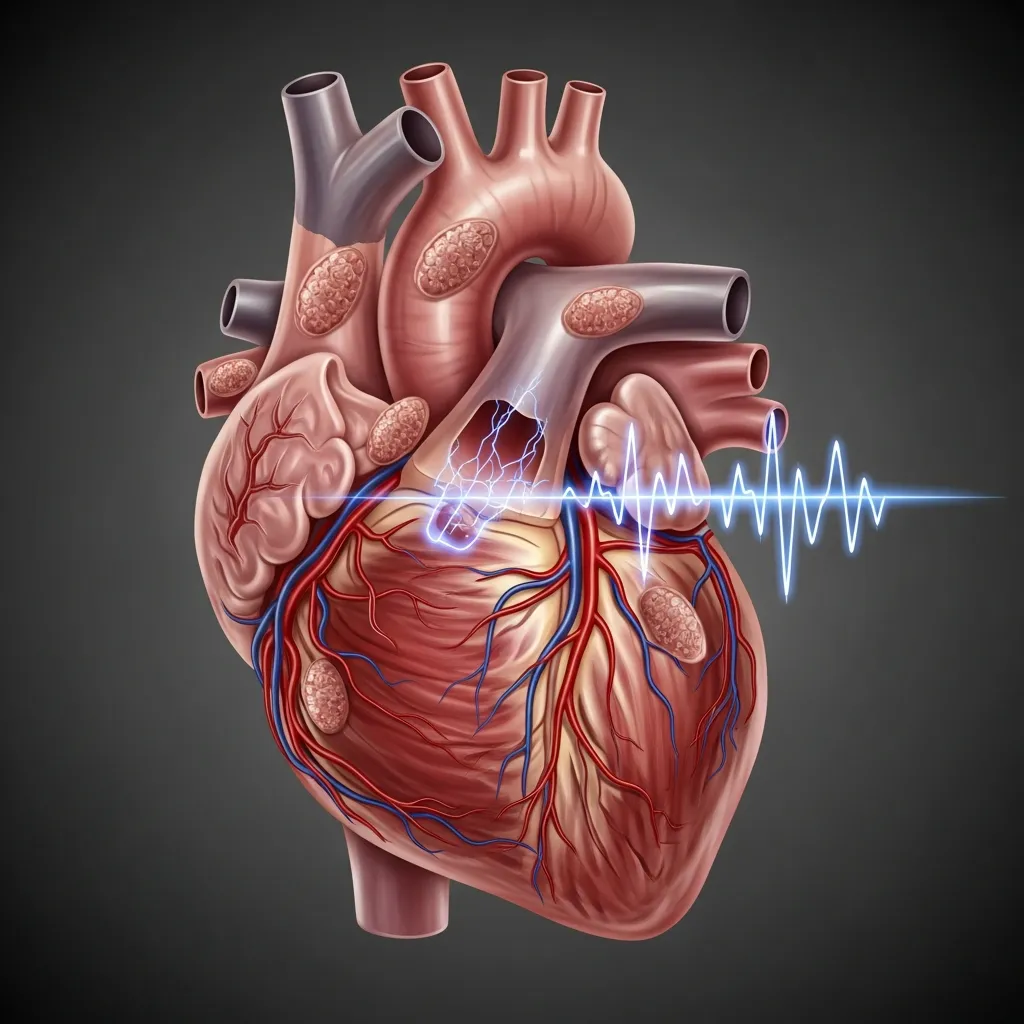 Heart complications are historically the leading cause of severe illness and mortality for patients with major forms of the disorder. When the body runs out of safe storage space for iron, the mineral deposits heavily into myocardial cells. These are the muscle cells responsible for the heart’s pumping action.
Heart complications are historically the leading cause of severe illness and mortality for patients with major forms of the disorder. When the body runs out of safe storage space for iron, the mineral deposits heavily into myocardial cells. These are the muscle cells responsible for the heart’s pumping action.
The presence of iron in these cells disrupts both the electrical and mechanical functions of the heart. Patients may experience arrhythmias, which are irregular heartbeats that feel like fluttering or racing. Over time, the toxic environment weakens the heart muscle, leading to cardiomyopathy and eventually heart failure. Some patients also develop pericarditis, an inflammation of the sac surrounding the heart.
Early detection is absolutely essential for cardiac health. Cardiologists use specialized imaging, specifically an MRI T2* scan, to measure the exact concentration of iron in the heart. Echocardiography is also used to evaluate how well the heart pumps blood. By catching iron buildup early, doctors can intensify chelation therapy to remove the iron before permanent damage occurs. This proactive approach significantly reduces the severe thalassemia complications in adults.
Liver Damage in Thalassemia
The liver is the primary storage site for excess iron in the human body. Because of this, it is usually the first organ to show signs of damage. As iron accumulates, it triggers chronic inflammation within the liver tissues. If this inflammation is not controlled, it leads to fibrosis, a condition where healthy tissue is replaced by scar tissue.
Without aggressive management, fibrosis can progress to cirrhosis, which is severe and irreversible scarring. A cirrhotic liver loses its ability to filter toxins, aid in digestion, and regulate blood clotting. Furthermore, patients with cirrhosis face a much higher risk of developing hepatocellular carcinoma, a type of primary liver cancer.
Another layer of risk comes from blood transfusions themselves. Before strict screening protocols were established in the 1990s, many patients contracted Hepatitis C or Hepatitis B from donated blood. Co-infection with these viruses accelerates liver damage significantly. Today, doctors monitor liver health meticulously using routine blood tests for liver enzymes, advanced MRI scans to quantify iron levels, and occasionally liver biopsies to assess the exact degree of scarring.
Endocrine System Dysfunction
The endocrine system is a network of glands that produce hormones regulating metabolism, growth, and reproduction. These glands are highly sensitive to iron toxicity.
The Pancreas and Blood Sugar
Iron deposition in the pancreas directly damages the beta cells responsible for producing insulin. This frequently leads to insulin resistance and diabetes mellitus. Patients must monitor their blood sugar levels carefully and often require insulin therapy to manage the condition.
Thyroid and Parathyroid Glands
When iron infiltrates the thyroid gland, it causes hypothyroidism. This results in extreme fatigue, unexplained weight gain, and a sluggish metabolism. Damage to the nearby parathyroid glands leads to hypoparathyroidism, which disrupts how the body regulates calcium. Low calcium levels can cause painful muscle cramps and tingling sensations in the extremities.
Pituitary and Adrenal Glands
The pituitary gland, located at the base of the brain, acts as the master control center for other hormones. Iron damage here frequently causes hypogonadism, leading to delayed puberty, restricted growth, and fertility issues. Additionally, damage to the adrenal glands can cause adrenal insufficiency. This means the body struggles to produce cortisol, a hormone vital for managing stress and maintaining healthy blood pressure. Endocrinologists treat these deficiencies with carefully tailored hormone replacement therapies.
Skeletal Abnormalities
 Because the bone marrow is constantly working overtime to produce red blood cells, it physically expands. This massive expansion thins out the hard outer layer of the bones, making them fragile and prone to fractures.
Because the bone marrow is constantly working overtime to produce red blood cells, it physically expands. This massive expansion thins out the hard outer layer of the bones, making them fragile and prone to fractures.
This condition is often compounded by endocrine issues, such as delayed puberty and low growth hormones, leading to severe osteoporosis. Patients face a heightened risk of breaking bones even from minor falls. The expansion of marrow also causes visible changes to the skeletal structure, particularly in the facial bones. This can result in a prominent forehead and misaligned teeth. In some cases, the body attempts to make blood outside the bones, a phenomenon called extramedullary hematopoiesis, which can cause masses that press against the spinal cord.
Management of bone health requires a comprehensive approach. Patients are often prescribed supplements like calcium and vitamin D. In cases of severe bone density loss, doctors may recommend bisphosphonates to help strengthen the skeletal structure and reduce fracture risks. General physical activity is encouraged to maintain mobility, though high-impact sports might need to be avoided depending on bone density scores. You can read more about anemia’s physical impact on the National Institutes of Health database.
Splenic Hyperactivity and Its Consequences
The spleen acts as a filter for the blood, removing old or damaged cells. In patients with this blood disorder, the red blood cells are structurally abnormal. The spleen recognizes these abnormalities and works aggressively to destroy them.
This constant heavy lifting causes the spleen to enlarge, a condition known as splenomegaly. An enlarged spleen often becomes overactive (hypersplenism). Instead of just removing bad cells, it begins trapping and destroying healthy red blood cells, white blood cells, and platelets. This worsens the patient’s anemia and increases their vulnerability to infections.
When the spleen becomes too large or active, surgeons may recommend a splenectomy to remove the organ. While this reduces the need for frequent blood transfusions, it removes a key part of the immune system. Patients who undergo this surgery require specific lifelong vaccinations and prophylactic antibiotics to protect against severe bacterial infections.
Kidney Complications
While less commonly discussed than heart or liver issues, the kidneys are also vulnerable. Chronic anemia forces the kidneys to filter blood at a much higher rate to maintain adequate oxygen delivery. This state of hyperfiltration places immense stress on the glomeruli, the tiny filtering units within the kidney.
Over decades, this stress can lead to glomerular and tubular dysfunction. The kidneys may begin leaking essential proteins into the urine or lose their ability to concentrate urine properly. Iron chelators, while necessary for survival, can also occasionally cause kidney irritation. Therefore, regular urine tests and blood tests tracking creatinine levels are vital to ensure the kidneys remain healthy.
Neurological Complications
Neurological issues are particularly relevant for patients with non-transfusion-dependent forms of the disease. Chronic anemia and a lack of oxygen to the brain can cause cognitive challenges and chronic headaches.
Furthermore, patients face an elevated risk of stroke. Abnormal red blood cells can increase blood clotting risks, leading to blockages in the brain’s blood vessels. Additionally, if extramedullary hematopoietic masses form near the spine, they can compress nerves, leading to pain, numbness, or loss of motor function. Prompt neurological evaluation is required if any of these symptoms appear.
Gastrointestinal Issues
The gastrointestinal tract often suffers secondary complications from the disorder. Chronic anemia prompts the body to absorb more iron from food, which can irritate the stomach lining.
Additionally, the rapid breakdown of abnormal red blood cells releases high amounts of bilirubin into the digestive system. The liver processes this bilirubin and sends it to the gallbladder. This excessive bilirubin frequently clumps together to form gallstones (cholelithiasis). Gallstones cause intense abdominal pain and may require surgical removal of the gallbladder to resolve.
Psychological and Quality of Life Aspects
Living with a chronic, life-threatening illness carries a heavy psychological burden. Frequent hospital visits for transfusions, daily medication schedules, and the constant fear of complications can take a severe toll on mental health.
Patients often experience anxiety, depression, and feelings of isolation. Children and teenagers may struggle with self-esteem issues due to delayed growth or noticeable facial bone changes. Comprehensive care must include access to social workers, psychologists, and support groups. Providing strong emotional support helps patients adhere to their challenging treatment regimens and improves their overall quality of life.
Advanced Therapies and Future Outlook
 The landscape of medical care is shifting rapidly, bringing new hope to families worldwide. For decades, the only treatments available were supportive, focusing on managing symptoms and delaying damage. Today, science is moving toward actual cures.
The landscape of medical care is shifting rapidly, bringing new hope to families worldwide. For decades, the only treatments available were supportive, focusing on managing symptoms and delaying damage. Today, science is moving toward actual cures.
Stem cell transplantation is currently the only established cure. By replacing defective bone marrow with healthy cells from a matched donor, patients can begin producing normal blood cells. However, finding a perfect match is difficult, and the procedure carries significant risks.
The most exciting frontier is gene therapy and CRISPR-Cas9 technology. Scientists are now able to extract a patient’s own stem cells, correct the faulty genes in a laboratory, and reintroduce them to the body. This eliminates the need for a donor and reduces the risk of rejection. Additionally, newer and more easily tolerated chelating agents are constantly being developed to make daily management easier. If you are exploring a diagnosis, reviewing a thalassemia test guide can help you understand the latest laboratory screening methods available.
Prevention and Early Diagnosis
Prevention and early detection remain the most effective strategies for managing public health regarding this genetic condition. Because the disorder is inherited, individuals with a family history should seek genetic counseling before planning a family.
A counselor can help determine if prospective parents carry the alpha thalassemia trait or beta mutations. If both parents are carriers, they have a 25 percent chance of having a child with a severe form of the disease. Prenatal diagnosis through amniocentesis can confirm the genetic status of a fetus early in pregnancy.
Furthermore, newborn screening programs have been implemented in many countries. By identifying the disorder immediately after birth, doctors can begin interventions before any tissue damage occurs. Recognizing alpha thalassemia symptoms early gives children the best possible chance at a healthy life.